Exploring the Role of Spring-Energized Seals in the Human Body: What You Didn’t Know About Medical Device Engineering
When we think of cutting-edge medical technology, most people imagine sophisticated diagnostics or robotic surgery. But the reality is that some of the most crucial components in life-saving medical devices are also the smallest, least visible ones—like seals. Specifically, spring-energized seals and PTFE-based seals are revolutionizing medical devices, especially those that are designed to be implanted in the human body.
In this blog, we’re going to take a deep dive into the world of seals used in medical implants. From ensuring fluid retention in insulin pumps to making sure heart pumps keep working reliably, the role these tiny components play in the human body is nothing short of extraordinary. Let’s explore what’s possible, and how these medical seals are advancing healthcare in ways many of us never knew about.
Sealing Solutions Inside the Human Body: A High-Stakes Engineering Challenge
Medical devices, especially those designed to be implanted or interact with the body, face extreme engineering challenges. These devices need to function in wet, dynamic, and corrosive environments, all while maintaining safety and longevity for the patient. Additionally, they must endure:
- Biocompatibility: Materials must not provoke immune responses or toxicity.
- Sealing for Life: Seals must be permanent and reliable to prevent fluid leakage, ensure that the device is securely housed, and protect delicate electronics from body fluids.
- Durability: Devices must endure the wear and tear of the human body’s movement—heartbeat, flexion, blood flow—all without degradation.
- Sterilization Resistance: Medical devices must withstand sterilization processes that could otherwise degrade or weaken seal integrity, especially in reusable devices.
This is where spring-energized seals made from advanced polymers like PTFE (Polytetrafluoroethylene) come into play. Unlike traditional elastomeric seals, which might degrade, swell, or harden under such conditions, PTFE-based seals are durable, chemically resistant, and biocompatible.
What Are Spring-Energized Seals?
Spring-energized seals are different from traditional seals due to their unique design. At their core is a metal spring, often made of stainless steel or other biocompatible materials, which is embedded in a PTFE or polymer seal body. This spring provides continuous radial or axial force to keep the seal in contact with the device’s moving or stationary surfaces.
In medical devices, these seals perform several critical functions:
- Maintain fluid retention in pumps and other devices.
- Ensure no external contamination enters delicate mechanical or electronic systems.
- Compensate for pressure fluctuations within the human body, like the natural expansion and contraction of tissues.
The combination of a spring’s constant pressure and PTFE’s resilience ensures that these seals maintain their integrity, even in the harshest environments.
Applications of Seals in Life-Saving Medical Devices
Let’s explore the specific applications where these advanced seals are making a life-saving difference:
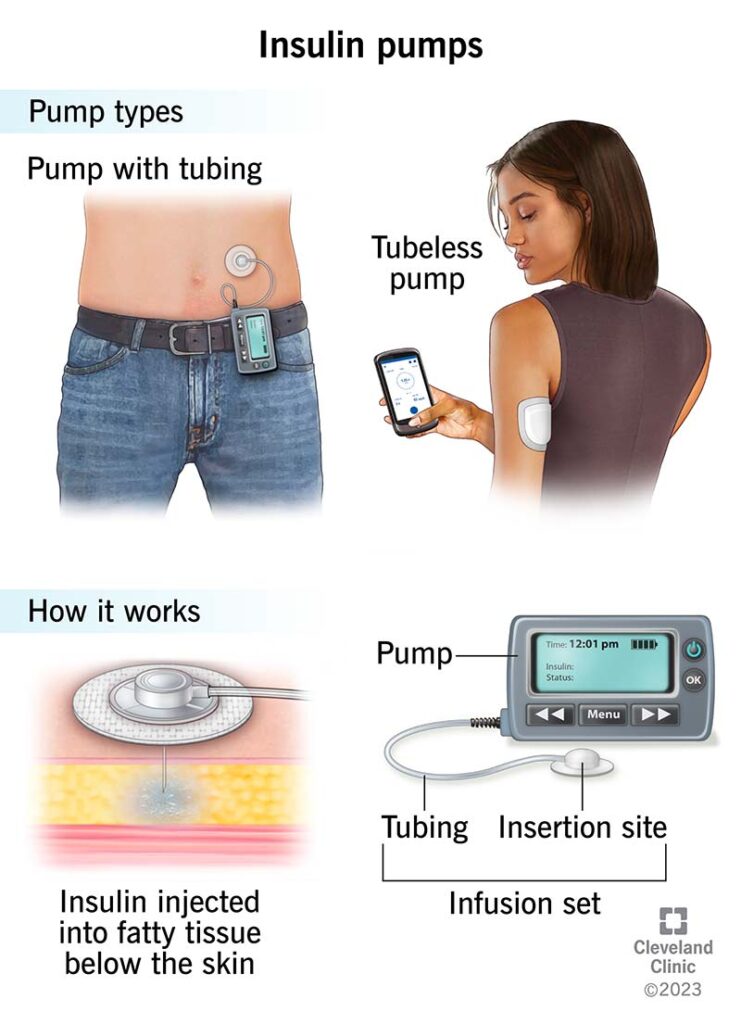
1. Insulin Pumps and Drug Delivery Systems
For patients with diabetes, insulin pumps are a life-saving device that delivers precise amounts of insulin to regulate blood sugar levels. These pumps rely on PTFE seals to ensure the device’s mechanical components, such as pumps and valves, operate smoothly and without leaks. The spring-energized seals used in these devices are critical because they must prevent insulin from leaking out while ensuring body fluids do not contaminate the device.
These seals must also endure the dynamic nature of insulin pumps, which are typically worn continuously by patients and subjected to movement, moisture, and temperature fluctuations.
In addition, PTFE’s chemical resistance makes it ideal for these devices, as it can resist breakdown or degradation in contact with insulin, the patient’s body fluids, and the various cleaning agents used for sterilization.
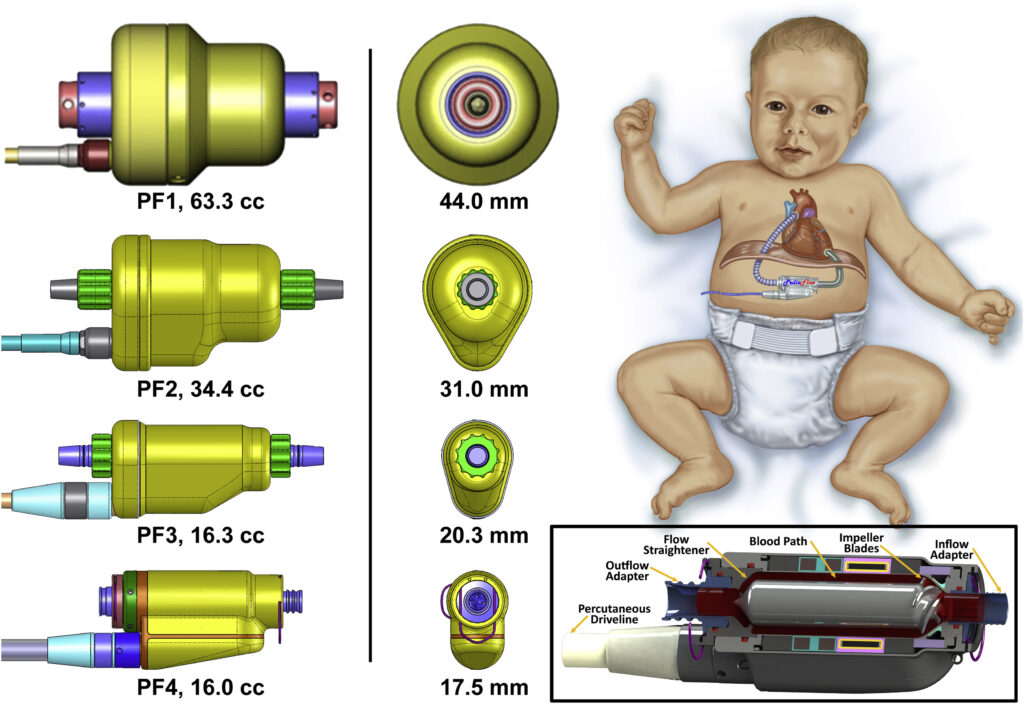
2. Implantable Heart Pumps and Artificial Hearts
The demands on seals in heart pumps are immense. The device must keep blood circulating without allowing leaks, while also enduring the constant rhythmic motion of the heart. The materials used in these seals must be biocompatible, chemically inert, and capable of withstanding high-speed fluid dynamics within the heart.
Spring-energized seals made from PTFE are ideal for this application because they maintain their sealing properties across a wide temperature range, resist blood corrosion, and remain flexible enough to handle the continuous motion without fatigue.
The continuous spring force also ensures that the seal remains intact even as the pump operates under different pressures, from normal blood flow to potential surges in the event of exertion or stress.
3. Prosthetics and Artificial Joints
In the case of advanced prosthetic limbs or artificial joints, seals play a crucial role in keeping these devices functional and long-lasting. Rotary seals, for instance, are used in joints to prevent debris and bodily fluids from entering and damaging the delicate internal mechanisms.
Since prosthetics are subjected to constant movement—bending, twisting, and rotating—traditional seals would quickly wear out. However, PTFE’s low friction properties mean that seals last longer, reduce wear on prosthetic joints, and maintain their functionality even under heavy and continuous use. This can be a game-changer for patients, allowing them more freedom of movement and longer intervals between device replacements or repairs.

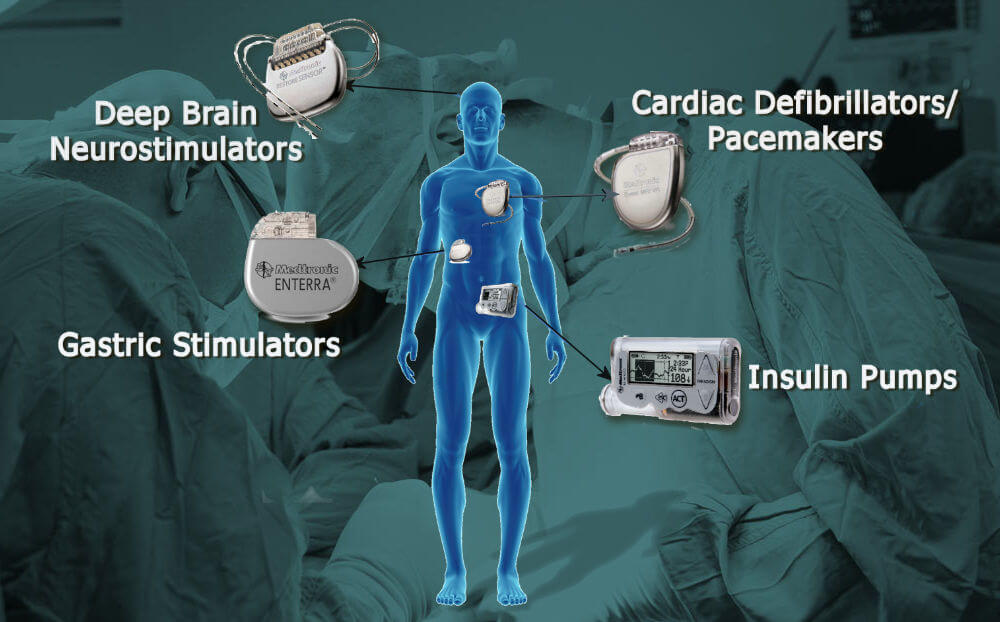
4. Pacemakers and Other Implantable Electronics
Pacemakers are vital for regulating a patient’s heartbeat, but the electronics inside must remain isolated from the body’s fluids to prevent corrosion and malfunction. Spring-energized seals provide a hermetic seal for the pacemaker’s enclosure, protecting its sensitive electronics from the moisture and salts found in the human body.
The seals used in these applications must be absolutely fail-safe, as any breach could cause the device to fail. PTFE and spring-energized designs offer an extra layer of protection by compensating for temperature and pressure variations inside the body, maintaining a constant seal under all conditions.
Why PTFE and Spring-Energized Seals Are Ideal for the Human Body
While there are other materials and seal designs available, PTFE spring-energized seals are uniquely suited to medical applications inside the human body. Here’s why:
- Biocompatibility: PTFE is FDA-approved and has been shown to be biocompatible, meaning it does not cause an immune response or other adverse reactions when implanted in the body. This is critical for long-term implantable devices.
- Chemical Resistance: PTFE seals are resistant to body fluids, medications, and sterilization chemicals, meaning they won’t degrade or break down in these harsh environments.
- Wide Temperature Range: Human body temperature may be constant, but devices must be able to withstand both the body’s natural environment and the high temperatures of sterilization. PTFE’s ability to operate in temperatures from -459°F to 500°F ensures the seal remains functional under all conditions.
- Low Friction: In applications like joint replacements or heart pumps, where movement is constant, PTFE’s low coefficient of friction ensures that seals maintain performance without adding wear to moving parts. This extends the lifespan of both the device and the seal.
The Future of Medical Seals: What’s on the Horizon?
The future of sealing technology in medical devices is promising, as advancements in materials science and engineering continue to evolve. New PTFE blends and spring technologies are being developed to enhance performance even further. For example, nanotechnology may allow for the creation of seals that are even more resistant to wear, while biodegradable polymers are being explored for temporary implants.
As medical devices become smaller, more efficient, and more complex, the role of sealing technology will only become more critical. Spring-energized seals are poised to continue their dominance in the medical sector, especially as we move toward more sophisticated implantable technologies, such as drug delivery microchips, brain-computer interfaces, and next-generation prosthetics.
“Small Seals, Big Impact”
While many may not realize it, seals are among the most critical components of life-saving medical devices. Spring-energized seals, particularly those made from PTFE, provide the reliability, durability, and safety necessary for these devices to function inside the human body. Whether it’s an insulin pump, a pacemaker, or a heart pump, these seals are often the unsung heroes ensuring that these devices work seamlessly and improve the lives of patients.
As medical technology continues to evolve, we can expect these seals to remain at the forefront, offering the precision and reliability needed for devices that will continue to push the boundaries of healthcare.
Contact Advanced EMC Technologies today to learn more about our cutting-edge solutions in medical sealing technology and how we can help bring your medical devices to the next level.


